Special Processes
While generally there are many ways to change the state a system, we often consider the following 4 special cases:
- Isobaric process - $P$ is fixed
- Isochoric (or isovolumetric) process - $V$ is fixed
- Isothermal process - $T$ is fixed
- Adiabatic process - $Q=0$, i.e. no heat exchange, thermally insulated
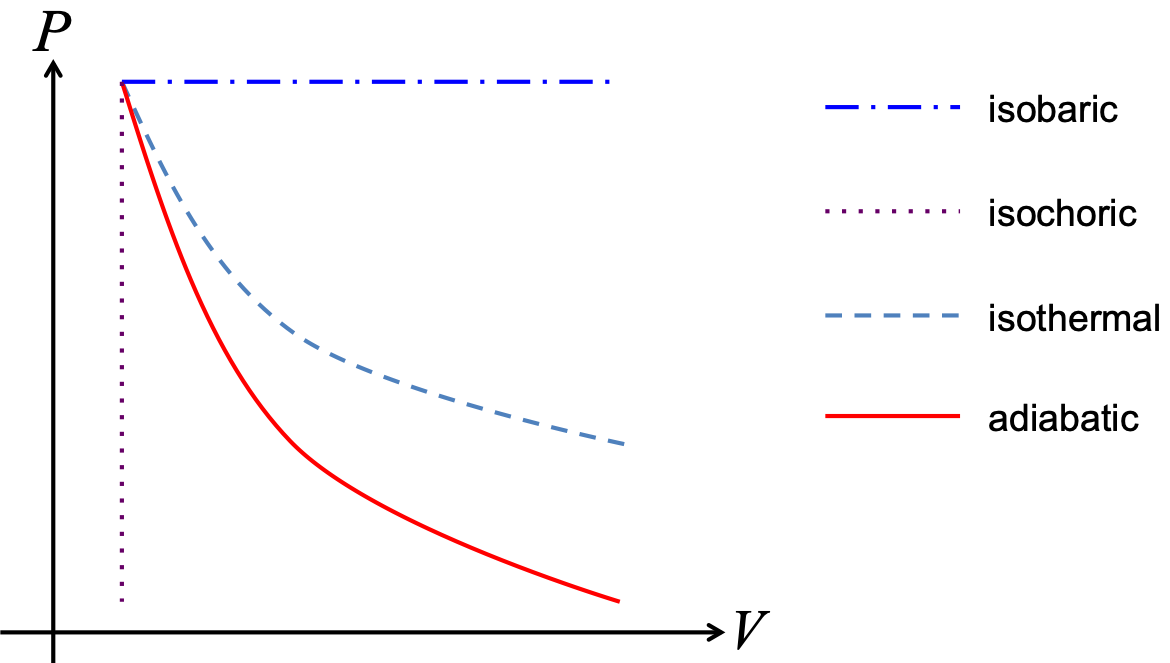
The isothermal curve is really just the ideal gas law $PV = nRT \Rightarrow P = \frac{nRT}{V}$, in other words $P\propto \frac{1}{V}$.
The adiabatic curve looks like an isothermal curve, except that it is slightly steeper. Mathematically, it is given by $P\propto \frac{1}{V^\gamma}$, where $\gamma = \frac{f+2}{f}$. The curve is also commonly written as $PV^\gamma = \text{constant}$.
