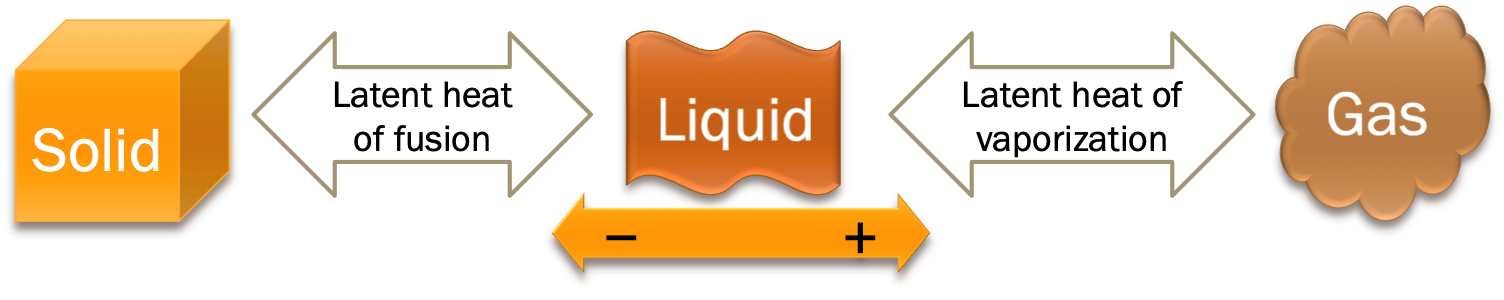
Heat is required during the change of phase, the amount of energy per unit mass is known as the latent heat. The basic equation is given by:
$$
Q = \pm m L
$$
The $\pm$ is needed because $Q$ is negative when a sample is losing heat (such as when water is being frozen into ice), but positive when it is gaining heat (such as when ice is melting into water). One must remember to use the correct sign when using this equation in computations.
There are two types of latent heat:
| Name |
Symbol |
Unit |
Meaning |
| Latent heat of fusion |
$L_f$ |
$ J/kg $ |
heat required to melt $1kg$ of sample at the melting point |
| Latent heat of vaporization |
$L_v$ |
$ J/kg$ |
heat required to vaporize $1kg$ of sample at the boiling point |
Latent Heat of Some Common Substances
| Substance |
$L_f (kJ/kg)$ |
Melting point ($^\circ C$) |
$L_v (kJ/kg)$ |
Boiling point ($^\circ C$) |
| Water |
334 |
0 |
2254 |
100 |
| Sulphur |
54 |
115 |
1406 |
445 |
| Mercury |
11 |
-39 |
294 |
357 |
| Nitrogen |
25 |
-210 |
199 |
-196 |
| Oxygen |
14 |
-219 |
213 |
-183 |
| Hydrogen |
60 |
-259 |
449 |
-253 |
Note that the unit in the table above is given in $kJ/kg$ instead of $J/kg$ because latent heat tends to be very big numbers in SI unit. In doing calculations, be sure not to forget the "kilo" in the unit or your calculations will all be off by a factor of 1000.
It is also worthwhile comparing the two equations for $Q$ you have encountered so far:
$$
\left\{
\begin{array}{l}
Q = mc\Delta T \text{ [no phase change, $\Delta T \neq 0$]}\\
Q = \pm mL \text{ [phase change, $\Delta T = 0$]}
\end{array}
\right.
$$
The absence of $\Delta T$ in the latent heat equation is based on the fact that temperature stays the same during a phase transition (i.e. $\Delta T=0$). For example, as water is boiling, the temperature stays at the boiling point of $100^\circ C$ no matter how strong the flame is.
For a related reason, there is no need to put $\pm$ in the specific heat equation because the sign of $Q$ in $Q=mc\Delta T$ is controlled by the sign of $\Delta T$. For example, when heat is supplied to a sample without phase change, temperature rises ($\Delta T\gt 0$), leading to $Q\gt0$. When it is being cooled, temperature drops ($\Delta T\lt 0$), leading to $Q\lt0$.
Try It Yourself (click to show)
Question will be loaded by load_exercise_example_all() defined in script_question.js
Question will be loaded by load_exercise_example_all() defined in script_question.js
Question will be loaded by load_exercise_example_all() defined in script_question.js
Try It Yourself (click to show)
Question will be loaded by load_exercise_example_all() defined in script_question.js
Question will be loaded by load_exercise_example_all() defined in script_question.js